Trusted Sterilization Information for Reprocessing Instruments with STERRAD™ Systems
About the STERRAD™ Sterility Guide
The STERRAD Sterility Guide (SSG) was founded to address an unmet need in the healthcare industry for reliable, up-to-date STERRAD Systems sterilization information. At ASP, we understand the importance of protecting patients and healthcare workers against hospital-acquired infections; and that’s been our mission: providing vital sterilization products and services where they’re needed most.
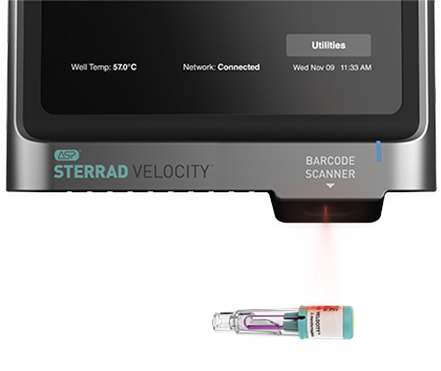
What makes the SSG different
We take great pride in our comprehensive process of adding devices to the SSG. ASP collaborates closely with device manufacturers, conducting rigorous testing and thorough evaluations to ensure every instrument listed meets validated dimensional and material claims for STERRAD Systems.
As pioneers in low-temperature sterilization, ASP also introduced the SSG - the first online tool of its kind of accessing validated device information for instrument reprocessing. Our commitment to innovation and reliability is why we confidently say that the SSG is the difference between assumed and assured.
We're here for you
The SSG is an easy-to-use online tool that acts as a bridge between the device manufacturer and central sterile departments globally.
Are you a medical device manufacturer?
Contact the Technical Business Solutions team at
[email protected]
to learn more about
our device validation testing program.
Our Values
Functionality
ISO 17644
ASP Provides
Functional Compatibility
Repeated-Cycle Testing
Sterility
ISO 14937
ASP Provides
Sterilization Efficacy
Sterilization Efficacy Testing
Reprocessing
ISO 17664
ASP Provides
SSG
Website
STERRAD® Systems sterilization information
